GMP or Good Manufacturing Practice guidelines provide guidance for manufacturing, testing and quality assurance as the respective manufacturer must meet the minimum requirements in the guidelines to assure that the products are of high quality and do not pose any risk or harm to the consumer or public.
Pharmaceutical, healthcare, chemical and food industry are some of the examples of industries that need to follow GMP guidelines. The cleanroom should be compliant with GMP EU Classification.
| Class | maximum particles/m3 | |||
| At Rest | At Rest | In Operation | In Operation | |
| 0.5 µm | 5 µm | 0.5 µm | 5 µm | |
| Grade A | 3,500 | 0 | 3,500 | 0 |
| Grade B | 3,500 | 0 | 350,000 | 2,000 |
| Grade C | 350,000 | 2,000 | 3,500,000 | 20,000 |
| Grade D | 3,520,000 | 29,000 | n/a | n/a |
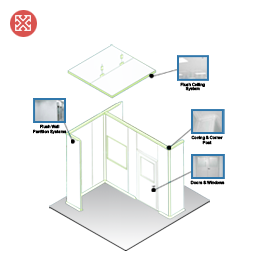
Recommended CLIN® systems for GMP compliant cleanroom:
• Ceiling: Frame Flush Ceiling System / “Z” Lock Flush Ceiling System
• Wall Partition: Frame Flush Wall Partition System / “Z” Lock Flush Wall Partition System
* comes with Coving and Pharma-type Corner Post
• Door: Pharma-type Swing Door / Sliding Door
• Lighting: Recessed Lighting
• Pass Box: Stainless Steel / EG Steel c/w Powder Coat
• Air Shower Unit: Stainless Steel / EG Steel c/w Powder Coat
• Filtration: HEPA Filter / ULPA Filter / Fan Filter Unit
Project Reference:
• B. Braun Medical Industries Sdn. Bhd.
• Haemonetics Malaysia Sdn. Bhd.
• Ambu Sdn. Bhd.